Morgenstern, J. Dynamic left ventricular outflow tract obstruction, First10EM, March 16, 2026. Available at:
https://doi.org/10.51684/FIRS.145254
Dynamic left ventricular outflow tract obstruction (LVOTO) is not a condition I learned about during residency, and it is not a condition I have diagnosed regularly, although I have probably missed it many many times. On First10EM, I usually prefer to start with a presenting symptom than a diagnosis, because that is how patients present. In fact, that is exactly how I started with LVOTO – in my approach to refractory shock (or the patient who isn’t responding to norepinephrine). However, unlike hypocalcemia or anaphylaxis, LVOTO seemed to require more explanation. It isn’t widely taught in emergency medicine circles, and requires some echocardiography skills that I am not really qualified to teach. Therefore, as an adjunct to the post on refractory shock, I thought I would share my notes on LVOTO.
What is dynamic LVOTO? (Some physiology and physics)
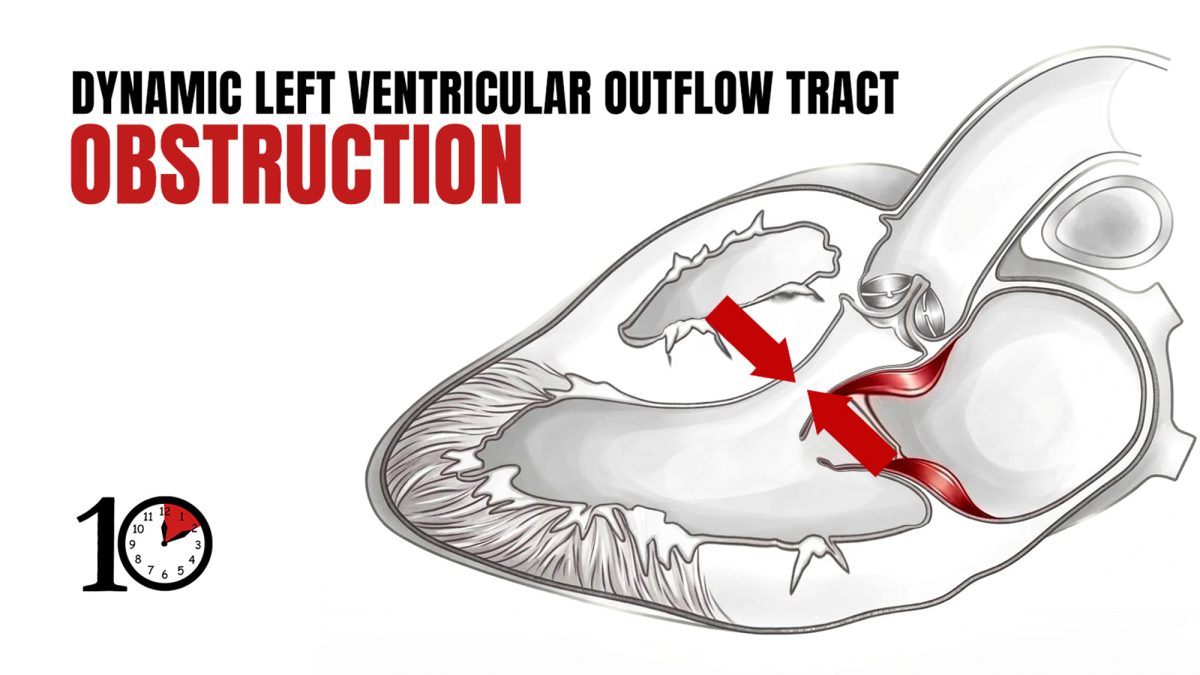
As the name implies, dynamic LVOTO is dynamic, varying with changes in preload, afterload, and contractility. (As compared to a fixed outflow tract obstruction like atrial stenosis). Fundamentally, it is the result of anterior motion of the mitral valve into the outflow tract during systole.
Do you remember Bernoulli’s principle? Basically, an increase in the speed of a fluid results in a decrease in pressure. When fluids go through a narrower space, they go faster (think putting your thumb over the end of a garden hose). So, when you have a narrow left ventricular out flow tract, blood will naturally flow faster. This is especially true when you are sick, and the heart is working harder. When a patient is tachycardic, there is less time in diastole for the left ventricle to fill, so narrow outflow tracts will get even narrower. When the afterload is low (such as in septic or distributive shock), the pressure gradient across the left ventricular outflow tract increases, and so flows become even faster. Rapid blood flow through the left ventricular out flow tract leads to low pressure, causing a vacuum effect that sucks the mitral valve into the tract. This leads to obstruction of flow, and often mitral regurgitation, ultimately decreasing cardiac output and exacerbating the shock state.
There will usually be an underlying anatomic abnormality. Hypertrophic cardiomyopathy is the most commonly discussed, with the enlarged septum narrowing the LVOT at baseline. However, there are many possible causes, not all of which are obvious when the patient is well. Friend and ultrasound expert Casey Parker tells me that ‘little old ladies with a “serpiginous” outflow tract who then get sick’ are the most common presentation he sees.
When to consider the diagnosis
LVOTO is on my checklist for patients with refractory shock. Anytime I am reaching for a second vasopressor, I am going to consider the diagnosis. It is a dynamic finding that can occur in any patient, but I am particularly suspicious if the patient has known cardiomyopathy or left ventricular hypertrophy. Acute right heart strain with dilatation (and therefore compression of the LV) is also a risk factor.
Clinically, obstruction of forward flow is likely to result in some degree of pulmonary edema. If you are good, you might also catch a new systolic murmur. Another hint that you might be dealing with LVOTO is that inotropes actually make the patient worse. I routinely reach for epinephrine as my second line vasopressor, but the increased heart rate and inotropy will exacerbate the LVOTO.
Regardless of risk factors, any patient with shock that is not improving on norepinephrine should probably have a bedside echo performed, and this is one of the key diagnoses to consider.
Let’s talk echo
Let me say upfront: I am not an ultrasound expert. Nowhere close. I am trying to summarize this ultrasound stuff to my level (or what I think will help the average community emergency doctor). I will include as many links as I can below to true ultrasound experts teaching with video examples – so I suggest getting the bulk of your education there.
In my first look at the heart, I am just going to be asking whether there is any reason to be thinking about LVOTO. Is there left ventricular hypertrophy? (If you want to measure, a basal septal wall thickness more than 1.2 cm puts you at risk.) Is the left ventricle adequately filling? (Left ventricular diameter at end-diastole of less than 2.2 cm.) Is the heart hyperdynamic?
If there is any concern, I will first focus on the mitral valve. This is probably easiest done using M mode, much like you would when measuring an EPSS. We are used to seeing the mitral valve hitting the septum at the end of diastole, but seeing the same movement during systole is pathologic.

The diagnosis is formally made by looking at gradients on continuous wave doppler, using an apical 5 chamber view. Obstruction is defined as a gradient more than 30 mmHg, but gradients greater than 50 are usually used as the clinical threshold for changing management. Although I am capable of generating this view, and can turn on the continuous wave doppler, ultrasound guru Casey Parker cautions that it is very easy to measure incorrectly (off axis), which would result in an underestimate of the velocity and false-negatives. His sense is that keen amateurs like me should try to qualitatively assess for the ‘dagger sign’ rather than worrying about specific numbers.
The dagger pattern: The classic pattern of LVOTO is described as a dagger on continuous doppler. It is a late peak, with a sudden drop off. The idea is that flow can start relatively normal, but as the anterior leaflet of the mitral valve is pulled into the outflow tract, and the outflow tract narrows, flow significantly increases. (This is a dynamic, rather than fixed, obstruction. The late peak in flow represents the dynamic process.) To really get a sense of this, you probably need to watch some of the videos linked below.


Management
Start with the basics. If there is any chance your patient is fluid depleted, start with IV fluids, which will improve preload and ventricular filling. If the patient is on after-load reducing medications (such as nitroglycerin in the context of pulmonary edema), discontinue those medications.
Avoid inotropes, and focus on pure vasoconstriction instead. Titrate off any inotropes that have already been started (epinephrine, dopamine, dobutamine), and transition to phenylephrine and/or vasopressin. Ensure that you are providing adequate sedation and analgesia, as endogenous catecholamines will have the same effect as the medications you are trying to titrate off.
If the patient is being ventilated, minimize PEEP to improve venous return.
The use of beta-blockers in a patient in shock seems contradictory, but tachycardia and significant inotropy both worsen LVOTO, and so beta-blockers may play an important role. I would probably be discussing this decision with my intensivist, and would definitely be using a short acting agent (esmolol).
Management of dysrhythmias: In the standard sepsis patient, I don’t actively manage atrial fibrillation. Unless it is extreme, I assume that the shock/sepsis is driving the tachycardia, and just focus on treating the underlying cause. In the setting of LOVTO, allowing time for the left ventricle to fill during diastole is essential, and so active management of dysrhythmias may be more important. If the arrhythmia is clearly new, try cardioversion. Otherwise, rate control with esmolol is again the treatment of choice.
Summary
If your patient is getting worse despite full dose vasopressors and/or inotropes, or as you are adding your second or third vasopressor, consider left ventricular outflow tract obstruction. These patients need an echo, and if you see a small hyperdynamic left ventricle, either get someone better at ultrasound than me, or focus on the outflow tract using M mode to see the anterior motion of the mitral valve during systole or continuous doppler to see the dagger pattern. Management is all about decreasing the speed of blood flowing through the outflow tract. Increase preload with fluids, slow the heart down to give it more time to fill during diastole, managing dysrhythmias, and transition from inotropic agents to pure vasoconstrictors.
OtherFOAMed
IBCC: Dynamic left ventricular outflow tract obstruction
Norepinephrine isn’t work – now what?? (Management of refractory shock)
EMCrit 420 – CV-EMCrit – Nuanced Presentations of Dynamic Left Ventricular Outflow Tract Obstruction (LVOTO): When It’s a “Two-Ventricle” Problem — and So Much More (My life would have been so much easier if this EMCrit episode had been posted a few months ago. Instead, I found it a few hours after my post went live. Honestly, I would skip my post and listen to the experts.)
References
Long B, Bridwell RE, Rapada R, Montrief T. Left ventricular outflow tract obstruction: A narrative review for emergency clinicians. Am J Emerg Med. 2025 Dec;98:153-159. doi: 10.1016/j.ajem.2025.08.052. Epub 2025 Aug 23. PMID: 40876217
Li X, Lin Y, Wang W, Zou Y, Zhang Z, Wu F, Shang J, Song Y, Liang M, Zhou Q. Dynamic left ventricular outflow tract obstruction in critical illness: etiology-driven management and the role of bedside transthoracic echocardiography. Crit Care. 2025 Nov 28;29(1):510. doi: 10.1186/s13054-025-05748-2. PMID: 41316420





3 thoughts on “Dynamic left ventricular outflow tract obstruction”
Great write up, just curious on your choice of when to start a second line pressor. You mention both in this article and in the refractory shock article, that you will consider alternative diagnosises when you reach for a second line vasopressor. When do you do that? You also mention in this article “full dose vasopressors” before adding a second line pressor. This is probably your local practice pattern, possibly due to local protocols on max doses of noradrenaline or similar rules. Just curious what these are, for you, as I don’t have the same protocols.
It’s a great question. I don’t actually have strict protocols about max doses, and really going into ‘higher than usual’ norepinephrine doses ends up prompting the same algorithm as starting a second pressor. It’s left vague intentionally, because the trigger is really the intuitive stop point described as the beginning of the other post: when things aren’t going as expected / when things don’t fit the pattern of 90% of the other patients you are seeing. Practically, if you wanted to push, if someone is needing more than 20 MCG/min of norepinephrine they probably need this checklist.